
Old and new subjects considered
through calculations of the dielectric
permittivity of water

Institute for Protein Research, Osaka University
Haruki Nakamura
(Molecular Scale WG)
Textbooks say that “water has approximately 80 times as high dielectric permittivity as a vacuum, and exhibits significant polarization.” Calculation of the dielectric permittivity of purified water using molecular simulation has had a long history, and some issues were clarified only recently. Looking back at these conventional and new issues might give researchers some points to keep in mind not only in a molecular simulation, but in an entire computational simulation. This paper describes an outline as follows.
The dielectric permittivity of a purified water system, ε is obtained from the statistical average of the sum total of the electric dipole moment![]() of individual water molecules
of individual water molecules![]() within the system, and is given as:
within the system, and is given as:
![]()
whereμ0 is the electric dipole moment of a water molecule, and GK is a scalar value called as the Kirkwood factor that is defined as:
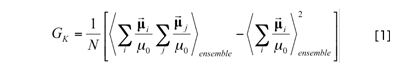
On the other hand, the distance-dependent Kirkwood factor given by the equation below serves as an important measure that reflects the alignment and structure of water molecules with extremely high sensitivity as compared to the radial distribution function (see Fig. 1 on the cover):
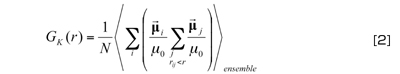
The factor shows a correlation with the electric dipole moment of water molecules on the first and second layers surrounding a water molecule, and even those on far-off layers. The second term of Equation [1] becomes zero in a sufficiently large statistical population, and therefore,
GK(r) given by Equation [2] wherer is infinite is equal to GK that represents the dielectric permittivity. In other words, a value on a sufficiently far-right point on the horizontal axis in Fig. 1 corresponds to the dielectric permittivity. It is calculated as approximately 96 by the particle-mesh Ewald (PME) method, a standard calculation technique, at 300 K and 1 atm under periodic boundary conditions. The discrepancy from 80 would have arisen from the TIP3P water molecule model1)used in the calculation.
Hydrogen-bond network relaxation in aqueous purified water is slow in terms of dynamics. Therefore, as shown in Fig. 2 (a) on the cover, simulation over a short period of time such as 1 to 2 ns is not enough for convergence of the dielectric permittivity value, and a long-time simulation at least 6 ns needs to be performed. The phenomenon, however, was pointed out in a systematic manner for the first time by Gereben and Pusztai (2011)2). This means that the values obtained by simulation over a short period of time in other papers published before are not reliable. My team had been faced with the same problem that the dielectric permittivity often varied in a short trajectory for several ns even using the same computational simulation method, and the finding by Gereben and Pusztai provided an unequivocal answer to our question. Furthermore, it was found that the second term of Equation [1] has not become a negligible, small amount of time average just for 1 to 2 ns as shown in Fig. 2 (b) on the cover. These findings are consequences through enhancement of computational resources.
In addition, there is another issue as regards handling long-range electrostatic forces in purified water. Only the interaction between water molecules within a certain cutoff distance
![]() had often been taken into consideration because of limited computational resources although the PME method is currently used as a standard technique. A simple idea is to set
had often been taken into consideration because of limited computational resources although the PME method is currently used as a standard technique. A simple idea is to set
![]() to as a large value as possible, but Yonetani3) pointed out in his paper that GK(r)
varied greatly by an order of magnitude in both positive and negative directions even if
to as a large value as possible, but Yonetani3) pointed out in his paper that GK(r)
varied greatly by an order of magnitude in both positive and negative directions even if
![]() was set to as large as 18Å.
The paper demonstrated that it not only caused a quantitative variation of the dielectric permittivity, but also failed to reproduce a qualitative water structure.
was set to as large as 18Å.
The paper demonstrated that it not only caused a quantitative variation of the dielectric permittivity, but also failed to reproduce a qualitative water structure.
In the past, Neumann4) proposed a method in which the artifact caused by the cutoff mentioned above is removed by the Reaction field method, assuming a dielectric material with the dielectric permittivity
![]() lies outside a sphere with a radius of
lies outside a sphere with a radius of
![]() , resulting in creation of a reaction field introduced by Fröhlich5). This method had been tried by many researchers, but has not been gaining high popularity recently, because it is difficult to estimate the parameter
, resulting in creation of a reaction field introduced by Fröhlich5). This method had been tried by many researchers, but has not been gaining high popularity recently, because it is difficult to estimate the parameter
![]() in a non-uniform system such as a protein solution. Recently, Dr. Ikuo Fukuda, RIKEN, proposed the “Zero-dipole summation principle” (ZD method)6)-8) as one of the non-Ewald methods. This is an outstanding method that incorporates the long-range effects by imposing not only the charge neutral condition proposed by Wolf9) but also that of electric dipole moment, allowing for high computational accuracy with a simple algorithm. By using the method, almost identical values of dielectric permittivity and GK(r)
as those from the PME method can be obtained as shown in Figs. 1 and 2, even though
in a non-uniform system such as a protein solution. Recently, Dr. Ikuo Fukuda, RIKEN, proposed the “Zero-dipole summation principle” (ZD method)6)-8) as one of the non-Ewald methods. This is an outstanding method that incorporates the long-range effects by imposing not only the charge neutral condition proposed by Wolf9) but also that of electric dipole moment, allowing for high computational accuracy with a simple algorithm. By using the method, almost identical values of dielectric permittivity and GK(r)
as those from the PME method can be obtained as shown in Figs. 1 and 2, even though
![]() is 12 Å to 14 Å. Namely, the ZD method works well even in a state where molecular interactions are cut off within a short range and only short-range interactions are taken into consideration. Interestingly, the ZD method gives exactly the same equation under certain conditions as that in the Reaction field method where
is 12 Å to 14 Å. Namely, the ZD method works well even in a state where molecular interactions are cut off within a short range and only short-range interactions are taken into consideration. Interestingly, the ZD method gives exactly the same equation under certain conditions as that in the Reaction field method where
![]() approaches infinity. It was also found that the ZD method has some characteristics in common with other various non-Ewald methods proposed recently8).It seems that common physics works as a basis in these methods in that no periodic boundary conditions are given.
approaches infinity. It was also found that the ZD method has some characteristics in common with other various non-Ewald methods proposed recently8).It seems that common physics works as a basis in these methods in that no periodic boundary conditions are given.
It is often the case in computational science that researchers compete with each other on computational speed according to given algorithms. This is definitely necessary, but creation of the original algorithms or models would open up completely new possibilities. My team has been working on studies that apply the ZD method mentioned above to heterogeneous systems such as protein and DNA solutions not as a periodic boundary system, but as a three-dimensional torus system, and are beginning to produce favorable results.
In computational research using new algorithms and models, researchers always conflict with reviewers who are insistent on the “Ptolemaic theory” and have some difficulties in publishing a paper. However, successful research results have a significant ripple effect. In fact, eliminating the use of the periodic boundary system in computation will allow us for easier high-speed simulation of many biological supramolecules with fewer computational resources.
Finally, my sincere acknowledgements are for the collaborators of this research, Dr. Ikuo Fukuda (RIKEN), Dr. Narutoshi Kamiya (Institute for Protein Research, Osaka University) and Dr. Yasushige Yonezawa (Kinki University).
【References】
1) W. L. Jorgensen et al., J. Chem. Phys . 79, 926 (1983); 2) O. Gereben, L.Pusztai, Chem. Phys. Lett. 507, 80 (2011); 3) Y. Yonetani, J. Chem. Phys. 124,204501 (2006); 4) M. Neumann, Mol. Phys. 50, 841 (1983); 5) H. Frohlich,“Theory of Dielectrics” Clarendon Press (1958); 6) I. Fukuda et al., J. Chem.Phys. 134, 164107 (2011); 7) I. Fukuda et al., J. Chem. Phys. 137, 054314 (2012);8) I. Fukuda, H. Nakamura, Biophys. Rev. 4, 161 (2012); 9) D. Wolf et al., J. Chem.Phys. 110, 8254 (1999)
BioSupercomputing Newsletter Vol.7
- SPECIAL INTERVIEW
- Interview with “K computer” Developer regarding Efforts in Exascale and Coming Supercomputer Strategies
Executive Architect, Technical Computing Solutions Unit, Fujitsu Limited
Motoi Okuda - Large-scale Virtual Library Optimized for Practical Use and Further Expansion into K computer
Professor, Department of Chemical System Engineering,
School of Engineering, The University of Tokyo
Kimito Funatsu
- Report on Research
- Old and new subjects considered through calculations of the dielectric permittivity of water
Institute for Protein Research, Osaka University
Haruki Nakamura
(Molecular Scale WG) - Development of Fluid-structure Interaction Analysis Program for Large-scale Parallel Computation
Advanced Center for Computing and Communication, RIKEN
Kazuyasu Sugiyama
(Organ and Body Scale WG) - SiGN : Large-Scale Gene Network Estimation Software with a Supercomputer
Graduate School of Information Science and Technology,
The University of Tokyo
Yoshinori Tamada
(Data Analysis Fusion WG) - ISLiM research and development source codes to open to the public
Computational Science Research Program, RIKEN
Eietsu Tamura
- SPECIAL INTERVIEW
- Understanding Biomolecular Dynamics under Cellular-Environments by Large-Scale Simulation using the “K computer”
Chief Scientist, Theoretical Molecular Science Laboratory,
RIKEN Advanced Science Institute
Yuji Sugita
(Theme1 GL) - Innovative molecular dynamics drug design by taking advantage of
excellent Japanese computer technology
Professor, Research Center for Advanced Science and Technology,
The University of Tokyo
Hideaki Fujitani
(Theme2 GL)
- Report
- Lecture on Computational Life Sciences for New undergraduate Students
HPCI Program for Computational Life Sciences, RIKEN
Chisa Kamada
