
Multi-scale Modeling of the Human
Cardiovascular System

Computational Science Research Program, RIKEN
Liang Fuyou
(Organ and Body Scale WG)
The cardiovascular system plays an important role in maintaining homeostasis; on the other hand, it is also highly vulnerable to diseases. Cardiovascular diseases are currently the number one killer of human beings. Research and technological development aimed to improve or support the diagnosis and treatment of circulatory diseases has been widely performed since decades ago. In particular, the correlations between vascular diseases and hemodynamic factors have been one of the most important issues.
The combination of medical imaging and fluid dynamics computing techniques makes it possible to noninvasively visualize in-vivo 3D-blood flows in a patient-specific manner, thus having been considered to carry some potential for clinical use. However, there are several critical problems with the application of the method. For example, the measurement of blood flow/pressure at the boundaries of a 3-D model is subject to various limitations related to measurement accuracy or vascular location suitable for the implementation of measurement. Moreover, the measurement can generally be done only when patients are in the resting state; as a consequence, the results of most hemodynamic computations reflect only the resting hemodynamic characteristics. As a matter of fact, hemodynamic characteristics are always changing in response to variations in physiological conditions.
Besides local blood flow patterns, hemodynamic phenomena viewed from a larger scale, such as systemic hemodynamic behavior, pulse wave propagation, carry medical value as well. For example, it is known that arterial pulse waves carry some characteristics that reflect the functional state of the heart and the morphological and mechanical properties of the arterial system. In fact, arterial pulse waves have long been used as a critical indicator in the diagnosis of cardiovascular diseases.
The complexity of the cardiovascular system raises another problem. For example, the cardiovascular system of a human is comprised by hundreds of arteries (veins), tens of millions of arterioles (venules), and billions of capillaries. These vessels are connected to form an extremely complex network structure. The complexity determines that it is impossible to construct a computational model that involves all the blood vessels since the data (such as those on vessel geometry and mechanical properties) for each vessel are not available, not to speak of the unaffordable computer resource required by such a model.
To handle the dynamic characteristics and the complexity of the cardiovascular system, we have developed a multi-scale modeling method. First, the microcirculation (including the capillaries, arterioles and venules), the arterial system and local large artery segments, are described respectively by a lumped-parameter (0-D), 1-D and 3-D model. Then, the models are coupled w ith each other to yield an integrated model of the entire cardiovascular system (Fig 1). The integrated model contains sub-models of different scales and hence is usually called a multi-scale model. An advantage of this modeling method is that it significantly reduces the computational cost for the whole model by representing the most complex microcirculation with a 0-D model that demands the least computer resource. From the point of view of hemodynamic analysis, the present model is preferable as well in that it allows computation of 3-D blood flows under various physiological/pathological conditions by coupling 3-D models of local blood flows with models of the remaining system.
The model has been applied to investigate various cardiovascular physiological phenomena related to hemodynamics. For instance, the modelbased simulations successfully reproduced the physiological phenomenon that the peak value increase and waveform variation of arterial pressure wave along the aorta are pronounced in young subjects but disappear with aging. More importantly, our model study indicated that such changes in arterial pressure wave propagation are caused by aging-associated arterial stiffening. We also coupled the model with an upper-arm cuff model and succeeded in evaluating the measurement accuracy of an automated sphygmomanometer and examining the validity of a device that assesses arterial stiffness based on the information obtained from an oscillometric cuff. Presently, we are developing methods to incorporate patient-specific clinical data (such as medical images for blood vessels, blood flow rate) into the model, aiming to preoperatively predict the postoperative outcomes or the risk associated with a surgery. In the future, by using a supercomputer, we will perform blood flow analysis that accounts for 3-D hemodynamic phenomena in a wider arterial region and incorporates more patient-specific information and physiological mechanisms.
[References]
[1] |
Liang F.Y., Takagi S., Himeno R., Liu H., Biomechanical characterization of ventricular-arterial coupling during aging: a multi-scale model study, J Biomech, 42:692-704, (2009) |
[2] |
Liang F.Y., Takagi S., Himeno R., Liu H., A computational model of the cardiovascular system coupled with an upper-arm oscillometric cuff and its application to studying the suprasystolic cuff oscillation wave concerning its value in assessing arterial stiffness. Comput Methods Biomech Biomed Engin. (in press) |
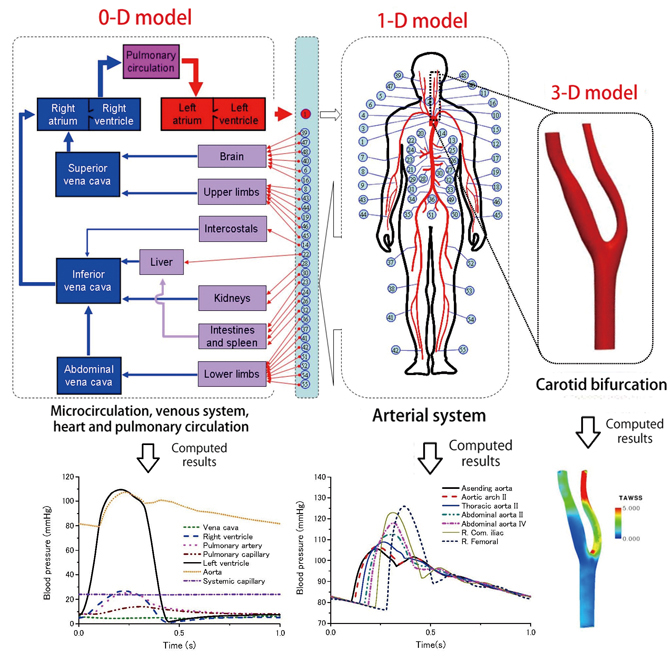
Fig 1 : Multi-scale modeling of the human cardiovascular system
(Upper panel: A schematic description of the model, Lower panel: Examples of the results computed with each sub-model)
BioSupercomputing Newsletter Vol.6
- SPECIAL INTERVIEW
- Development of New Fluid-structure Interaction Analysis (ZZ-EFSI) Resulting in Rapid Achievement of High Operation Performance
Research Associate Professor, School of Engineering, The University of Tokyo Kazuyasu Sugiyama - Interview with High-performance Computing Team Members: Continued Efforts in Tuning to Harness the Potentials and the High Capability of the K Computer
Group Director of Computational Molecular Design Group,
Quantitative Biology Center, RIKEN
Makoto Taiji
Senior Researcher of High-performance Computing Team,
Integrated Simulation of Living Matter Group,
Computational Science Research Program, RIKEN
Yousuke Ohno
Senior Researcher of High Performance Computing Development Team,
High Performance Computing Development Group,
RIKEN HPCI Program for Computational Life Sciences
Hiroshi Koyama
Researcher of High-performance Computing Team,
Integrated Simulation of Living Matter Group,
Computational Science Research Program, RIKEN
Gen Masumoto
Research Associate of High-performance Computing Team,
Integrated Simulation of Living Matter Group,
Computational Science Research Program, RIKEN
Aki Hasegawa
- Report on Research
- Functional Analysis of Multidrug Efflux Transporter AcrB by All-Atom Molecular Dynamics Simulation
Graduate School of Nanobioscience,
Yokohama City University
Tsutomu Yamane,
Mitsunori Ikeguchi
(Molecular Scale WG) - Multi-scale Modeling of the Human Cardiovascular System
Computational Science Research Program,
RIKEN Liang Fuyou (Organ and Body Scale WG) - Toward a spiking neuron-level model of the early saccade visuomotor system
Kyoto University Jan Moren
Nara Institute of Science and Technology Tomohiro Shibata
Okinawa Institute of Science and Technology Kenji Doya
(Brain and Neural Systems WG) - Developing the MD Core Program for Large Scale Parallel Computing
Computational Science Research Program,
RIKEN Yousuke Ohno (High-performance Computing Team)
- SPECIAL INTERVIEW
- Pioneering the Future of Computational Life Science toward Understanding and Prediction of Complex Life Phenomena
Program Director of RIKEN HPCI Program for Computational Life Sciences
Toshio Yanagida
Deputy- Program Director of RIKEN HPCI Program for Computational Life Sciences
Akinori Kidera
Deputy- Program Director of RIKEN HPCI Program for Computational Life Sciences
Yukihiro Eguchi
- Report on Research
- Free Energy Profile Calculations for Changes in Nucleosome Positioning with All-Atom Model Simulations
Quantum Beam Science Directorate, Japan Atomic Energy Agency
Hidetoshi Kono, Hisashi Ishida, Yoshiteru Yonetani, Kimiyoshi Ikebe (Field 1- Program 1) - Estimation of Skeletal Muscle Activity and Neural Model of Spinal Cord Reflex
Information Science and Technology, The University of Tokyo
Yoshihiko Nakamura (Field1 - Program 3)
- Report
- ISLiM Interim Accomplishment Meeting in 2011
Computational Science Research Program, RIKEN Eietsu Tamura - Computational Life Sciences Classes Held in High Schools
HPCI Program for Computational Life Sciences, RIKEN
Chisa Kamada, Yasuhiro Fujihara, Yukihiro Eguchi
