
Simulation for Charged
Particle Therapy

Organ and Body Scale Team
Kenichi L. ISHIKAWA
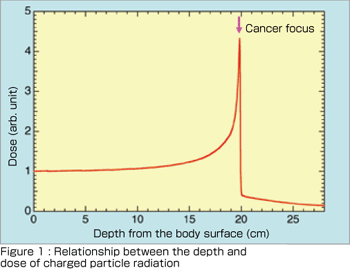
Charged particle cancer therapy is an advanced radiotherapy to cure cancer noninvasively (without surgery) by intensively irradiating the tumor with a charged particle beam (a beam generated by accelerating ions such as carbon ions). X-rays used in conventional radiotherapy do not stop at the tumor and go through the body, thereby uniformly affecting the tissue along their paths. In contrast, a charged particle beam deposits maximum energy immediately before it stops, as shown in Figure 1. Thus, a charged particle beam with an appropriately chosen energy stops exactly at the cancer focus to attack the tumor in a pinpoint manner. Charged particle therapy is presently approved as a highly advanced medical technology and is attracting increasing attention.
When performing a surgical operation, a doctor can visually confirm the area for resection, but during charged particle therapy, it is impossible to monitor which area is being irradiated. Thus, it is required beforehand to calculate the desired condition of the beam irradiation so that the radiation dose (the amount and the effect of the radiation) can be efficiently concentrated on the cancer focus. Hence, the quality of therapy can be further improved by sophisticated simulation.
A charged particle beam collides with atoms in the human body and causes various kinds of phenomena, such as ionization and nuclear reaction. In a human body, composed of different kinds of tissues, how and where the beam reacts is complex and stochastic. Based on the Monte Carlo simulation, which uses random numbers to trace the transport and reaction of numerous incident particles in its entirety as much as possible, we are endeavoring to develop a method to calculate the dose of charged particle radiation more accurately.
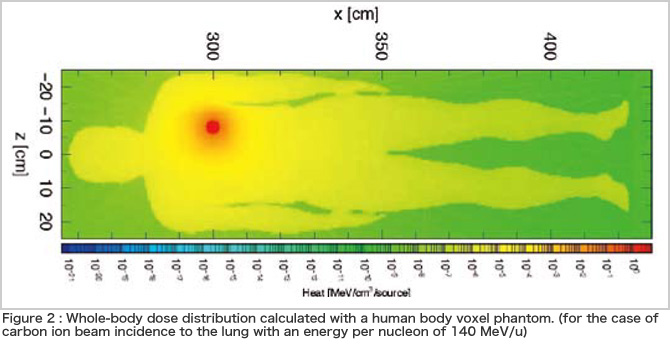
In the current therapy, calculations usually cover only the effect on the cancer focus and its periphery. Since a charged particle beam is a type of radiation, however, it can cause secondary cancer years or decades after recovery from the initial cancer. It is an irony that the therapy has become so effective that the treatment planning now has to take account of the risk of secondary cancer that the therapy itself may cause several decades later (how greedy humans are!). For that purpose, we are conducting research on how to calculate the dose distribution of charged particle beams in the entire human body. Figure 2 illustrates an example of the results of the whole-body dose calculation using a voxel phantom, which represents the body as a group of small cubes (voxels). Through the calculation mentioned above, our simulator can determine the type of reaction or particle that contributes to effectively delivering a dose to areas far from the cancer focus. Thus, the simulator is expected to provide safer therapy with a lower risk.
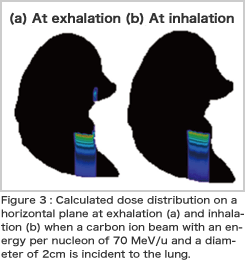
The human body moves during respiration, but even so, if CTs are taken repeatedly, the amount of X-ray exposure may be increased. To overcome this problem, we are researching the effect of respiration by simulating the movement of the lung by computers. By utilizing the simulation coupled with the model of lung motion (the spring network model) developed by Prof. Shigeo Wada's group at Osaka University, we calculated how a dose of charged particle radiation is affected by respiration. The calculation results are shown in Figure 3. The dose distribution at inhalation differs from that at exhalation. It is expected that more precise therapy is realized if such effects are taken into consideration.
The simulation we are working on requires a huge amount of calculation. We are dreaming to develop a system capable of elaborate treatment planning on the entirely computational basis from a single CT scanning, with account of the movement of organs and the risk of secondary cancer, using the nextgeneration supercomputer.
BioSupercomputing Newsletter Vol.1
- SPECIAL INTERVIEW
- Innovative Approach for Understanding Phenomena of Life Exploring New Possibilities with Bio-supercomputing
Computational Science Research Program Deputy Program Director Ryutaro HIMENO
- A Message from the Team Leader
- Simulations to Understand the Functions of the Biopolymers that Play Fundamental Roles in Life
Molecular Scale Team Team Leader Akinori KIDERA - Develop a 3-D Model of the Entire Human Body and Understand In Vivo Phenomena to Utilize for Medical Purposes
Organ and Body Scale Team Team Leader Shu TAKAGI - The Fourth Methodology (Data Analysis Fusion): Transforming Biology into a Predictable Science
Data Analysis Fusion Team Team Leader Satoru MIYANO
- Report on Research
- Prediction of Transmembrane Dimer Structure of Amyloid Precursor Protein using Replica-Exchange Molecular Dynamics Simulations
Molecular Scale Team Naoyuki MIYASHITA / RIKEN Advanced Science Institute (Molecular Scale WG) Yuji SUGITA - Simulation for Charged Particle Therapy
Organ and Body Scale Team Kenichi L. ISHIKAWA - Prospects of Prognostic Prediction Based on Genome-wide Association Study and Genetic/Non-genetic Factors
Riken Center for Genomic Medicine (Data Analysis Fusion WG) Naoyuki KAMATANI - Key Technology Supporting Petascale Computing
High-performance Computing Team Kenji ONO / Satoshi ITO / Daisuke WATANABE
