
Multi-scale, multi-physics
heart simulator UT-Heart
Graduate School of Frontier Sciences, the University of Tokyo
(From the left) Toshiaki Hisada, Seiryo Sugiura,Takumi Washio, Jun-ichi Okada, Akihito Takahashi
(Organ and Body Scale WG)





The human heart comprises multi-physics problems that widely cover various electric (ion current, excitation propagation, electrocardiogram, etc.), chemical (mass transport, reaction, energy conversion, etc.), and mechanical (myocardial tension, blood pressure, blood flow, etc.) phenomena originated from the biochemical reaction by ATP (adenosine triphosphate) hydrolysis as an energy source. In terms of spatial scale, it comprises multi-scale problems that range from protein molecules (up to 10 nm) to organs (up to cm) via cells (up to 100μm) and tissues (up to mm), and finally ejection of blood is achieved.
ATP is consumed as energy for the relative sliding movement between the numerous actin filaments and myosin filaments that comprise 50 to 60 myofibrils present in a single cardiac muscle cell. It is known that the relative sliding of the filaments results from the tractive force that is produced by the myosin molecule heads extending from the myosin filaments combining with the actin filaments. Hypotheses for the specific mechanism involved are however diverse, and still remain under discussion. Figure 1 (right) shows a hypothesis of the rotation of myosin heads. In the meantime, cardiac hypertrophy is generally interpreted as a phenomenon where the wall thickness (myocardial cross section) increases to cope with a situation of high blood pressure such as valvular diseases and hypertension. In a disease called hypertrophic cardiomyopathy, however, the heart wall is thickened for no reason at normal blood pressure. In an initial study, the thickening had been interpreted as occurring to compensate for degrading cardiac function due to point mutation of myosin molecules (represented by mutation of the 403rd amino acid at which the myosin molecule binds with the actin molecule). In a later study, however, it was reported that the mutated myosin is rather hyperactive[1]. Then, what happens to the heart by admixture with the hyperactive myosin?
We have developed cardiac multi-scale analysis technologies on massively parallel computers so far[2], and are earnestly addressing this issue using the “K computer”. More specifically, we had inevitably tried to solve the probabilistic binding state of actin molecules and myosin molecules by conceiving an average molecule, and applying it to an equation expressing the state transition in the past, due to limitations of computer power[3]. However, we came to be able to seamlessly solve the tissue and organ movements through cell contraction by starting with a concrete simulation of the probabilistic movements of the individual myosin heads with intramolecular elastic elements modeled on a spring as shown in Fig. 1, by taking advantage of the characteristics of massively parallel computers. The myosin head movement has a characteristic, known as “Cooperativity,” whereby binding of a certain myosin head with an actin filament encourages its neighboring myosin heads to bind with the actin. This computation model enables the introduction of such a characteristic as it is, and thus the analysis of heartbeat in the case of admixture with the previously-mentioned hyperactive myosin. The following specific procedures will be undertaken:
(1) Simulate the probabilistic micro-scale behavior of myosin molecules with cooperativity directly by the Monte Carlo method;
(2) Embed the above-mentioned Monte Carlo model in the muscle fiber portion of the mesoscale cell structural element model including the intercellular space and so on to properly address the dynamic interactions between scales physically; and
(3) Bind the movements of the above secondary structural element model and the macro-scale organ model by the homogenization method.
The realization of the above analytical method enables the simulation-based analysis of such issues as what effects are produced by the molecular level state change law and cellular tissue level structural elements on heartbeat performance and energy efficiency and, conversely, what effects are produced by the feedback of macromuscular contraction and relaxation on the molecular level state change. Such a simulation platform may also serve as a means for molecular biologists to verify their own hypotheses from a new perspective in future.
The research and development and validation of the UT-Heart are jointly implemented by the Fujitsu Limited Next Generation Technical Computing Unit (a group led by Yoshimasa Kadooka, General Manager and Head, Application Research and Development Division), and the University of Tokyo Hospital (a group led by Visiting Professor Ryozo Nagai). The clinical study was approved by the Institutional Review Board at the University of Tokyo.
[References]
[1] Belus A, Piroddi N, Scellini B, Tesi C, Amati GD, Girolami F, Yacoub M, Cecchi F, Olivotto I, Poggesi C, The familial hypertrophic cardiomyopathy-associated myosin mutation R403Q accelerates tension generation and relaxation of human cardiac myofibrils, J Physiol, 586(Pt 15), 3639-3644, 2008
[2] Hosoi A, Washio T, Okada J, Kadooka Y, Nakajima K, Hisada T, A Multi-Scale Heart Simulation on Massively Parallel Computers, SC10, NewOrleans, proceedings 1-11, 2010
[3] Washio T, Okada J, Sugiura S, Hisada T, Approximation for Cooperative Interactions of a Spatially-detailed Cardiac Sarcomere Model, Cellular and Molecular Bioengineering, 5, 113-126, 2012
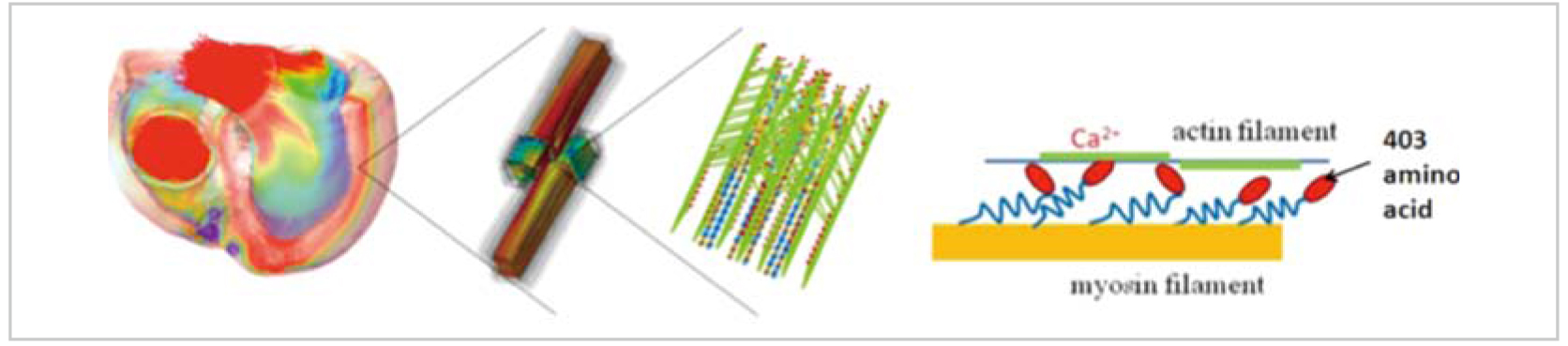
Fig. 1: K computer-based Multi-scale Heartbeat Simulation from Sarcomere Kinetics
BioSupercomputing Newsletter Vol.8
- SPECIAL INTERVIEW
- Grand Challenge opens the way to the future of life science through innovative approach
Program Director, Computational Science Research Program, RIKEN
Koji Kaya - A Landmark Project that Brought on an Innovation in the Field of Life Scienc
Deputy-Program Director, Computational Science Research Program, RIKEN
Ryutaro Himeno
- Report on Research
- Multi-scale, multi-physics heart simulator UT-Hear
Graduate School of Frontier Sciences, the University of Tokyo
Toshiaki Hisada, Seiryo Sugiura, Takumi Washio,
Jun-ichi Okada, Akihito Takahashi
(Organ and Body Scale WG) - Simulation Model for Insulin Granule Kinetics in Pancreatic Beta Cells
Graduate School of System Informatics, Kobe University
Hisashi Tamaki(Cell Scale WG) - The road to brain-scale simulations on K
Brain Science Institute, RIKEN, Institute of
Neuroscience and Medicine (INM-6),
Juelich Research Center
Medical Faculty, RWTH Aachen University Markus Diesmann
(Brain and Neural Systems WG) - MD Core Program Development for Large-scale Parallelization
Computational Science Research Program, RIKEN
Yousuke Ohno(High-performance Computing Team)
- SPECIAL INTERVIEW
- Aiming to realize hierarchical integrated simulation in the circulatory organ system
and the musculoskeletal / cerebral nervous systems
Professor, Department of Mechanical Engineering and Department of Bioengineering The University of Tokyo
Shu Takagi(Theme3 GL) - Leading-edge large-scale sequence data analysis with K computer in order to promote the understanding of life programs and their diversity
Professor, Human Genome Center, The Institute of Medical Science, The University of Tokyo
Satoru Miyano(Theme4 GL)
- Report
- 4th Biosupercomputing Symposium Report
Computational Science Research Program, RIKEN
Eietsu Tamura - “K Computer” Compatible Computer: Installation of SCLS Computer System
HPCI Program for Computational Life Sciences, RIKEN
Yoshiyuki Kido
